What we investigate
Our laboratory studies the molecular and cellular mechanisms of tissue repair, with particular emphasis on the roles of growth factors and their downstream signalling pathways in this process. In particular, we are interested in the parallels between wound healing and cancer at the cellular and molecular level.
KEYWORDS
activin, atopic dermatitis, FGF, skin cancer, wound healing
Our research in more detail
Tissue injury initiates a series of events, which lead to at least partial reconstruction of the injured body site. With the exception of the liver, which can completely regenerate in most cases, repair of other organs is imperfect and results in scar formation with functional impairments. There are many conditions in humans, which are associated with impaired tissue repair, including old age, steroid treatment, diabetes and cancer. Therefore, there is a strong need to improve the healing process. This requires a detailed understanding of the underlying cellular and molecular mechanisms. By trying to elucidate these mechanisms, our research shall help to develop new strategies for the improvement of tissue repair.
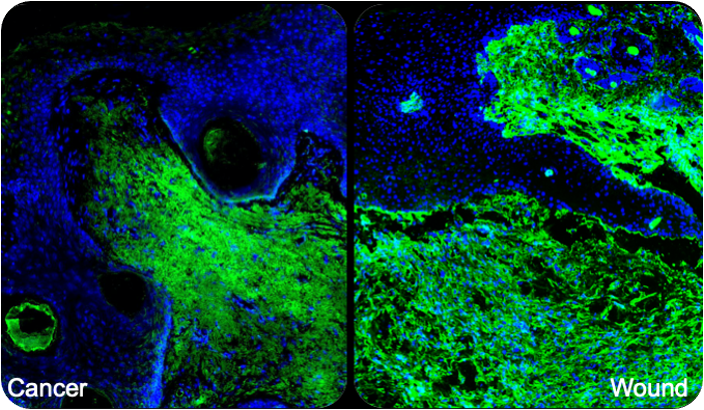
An exciting aspect of our research is the analysis of the parallels between tissue repair and cancer. We identify and functionally characterize genes and signaling pathways, which orchestrate both processes, with a focus on the role of growth factors and transcriptional regulators. We use the mouse as a model organism to address these questions. Collaboration with clinical partners will help to determine the importance of our findings for the human situation and to transfer our research results into clinical practice.Selected publications
SKINTEGRITY.CH Principal Investigators are in bold:
- Wietecha M*, Lauenstein D, Rion S, Jin J, Goppelt A, Claassen M, Levesque MP, Dummer R and Werner S* (2023). Phase-specific signatures of wound fibroblasts and extracellular matrix patterns define cancer-associated fibroblast subtypes and predict cancer outcome. Matrix Biol. 119, 19-56. *Corresponding authors.
- Cangkrama M*, Liu H, Whipman J, Zubair M, Matsushita Azuma M, Kopf M, Innocenti M*, and Werner S* (2022). A pro-tumorigenic mDia2-MIRO1 axis controls mitochondrial positioning and function in cancer-associated fibroblasts. Cancer Res. 82, 3701-3717. *Corresponding authors.
- Wietecha M+, Pensalfini M+, Müller B, Cangkrama M, Brinckmann J, Mazza M* and Werner S* (2020). Activin-mediated reprogramming of the fibroblast transcriptome and matrisome controls the biomechanical properties of skin wounds. Nat Commun, 11, 2604. +Joint first authors, *corresponding authors.
- Hiebert P, Wietecha M, Cangkrama M, Haertel E, Mavrogonatou E, Stumpe M, Steenbock H, Grossi S, Beer H-D, Angel P, Brinckmann J, Kletsas D, Dengjel J, and Werner S (2018). Nrf2-mediated fibroblast reprogramming drives cellular senescence by targeting the matrisome. Dev Cell 46, 1-17.
- Seltmann K+, Meyer M+, Sulcova J, Kockmann T, Wehkamp U, Weidinger S, auf dem Keller U*, and Werner S* (2018). Humidity-regulated CLCA2 protects the epidermis from hyperosmotic stress. Sci Transl Med 10, eaao4650. +Joint first authors, *corresponding authors.
