What we investigate
The research interests of my laboratory lie at the crossroads of immunology and vascular biology. The main aims are to elucidate basic mechanisms of leukocyte migration through afferent lymphatic vessels, lymphatic remodeling under pathologic conditions and the immunomodulatory functions of lymphatic endothelial cells.
KEYWORDS
time-lapse confocal / multiphoton microscopy, leukocyte trafficking, immune modulation, lymphatic vessels, lymphatic flow, lymphatic endothelial cell phenotype, single-cell sequencing
Our research in more detail
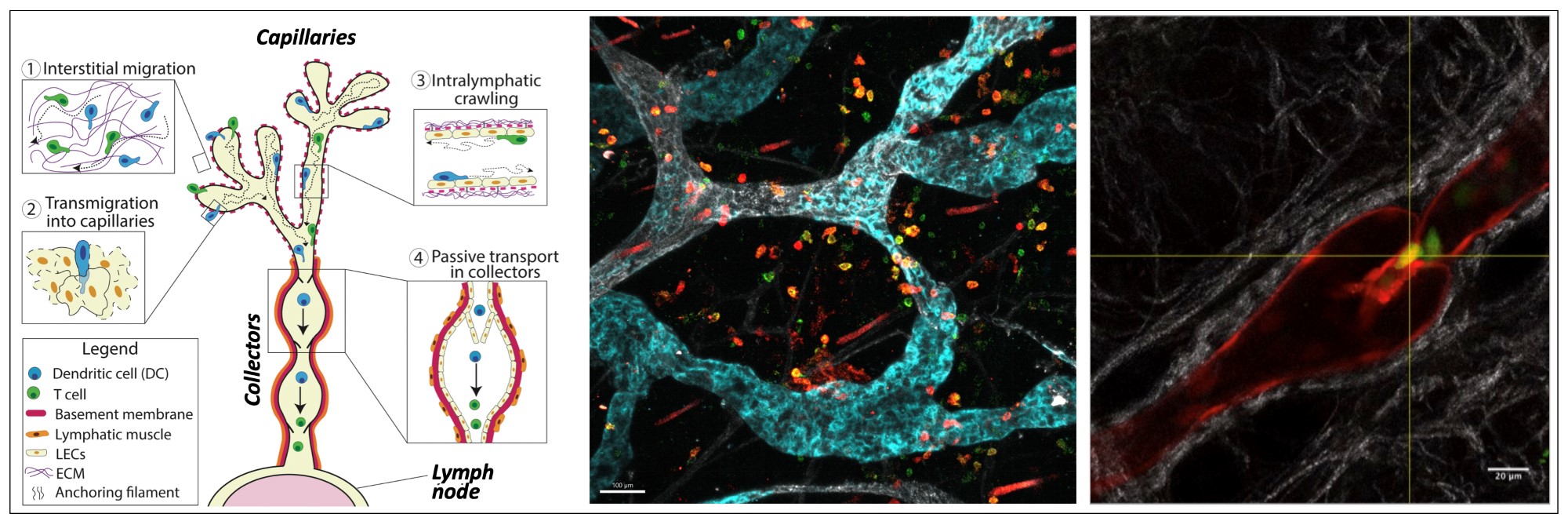
Afferent lymphatic vessels are present in virtually all tissues of the body and well known for draining excess tissue fluid and maintenance of fluid homeostasis. By transporting leukocytes, in particular antigen-presenting dendritic cells (DCs), inflammatory mediators and soluble antigens to draining lymph nodes, afferent lymphatic vessels additionally play an important role in the induction and regulation of adaptive immunity. Initially considered to be inert tubes, it is nowadays clear that lymphatics play a much more active role in immune induction and the resolution of inflammation. For example, lymphatic endothelial cells (LECs) actively regulate leukocyte migration by upregulating adhesion molecules and chemokines under inflammatory conditions, and the lymphatic network expands and remodels in disease conditions (e.g. chronic inflammation, infection or cancer). Moreover, in recent years it was found that LECs of afferent lymphatics and in lymph nodes express immune-modulatory molecules that affect the activity of DCs or of T cells. LECs are nowadays considered to mainly exert immune-dampening functions and to be relevant for preventing auto-immunity and for the maintenance of tolerance.
We employ imaging- and transcriptomics-based approaches to investigate leukocyte migration through afferent lymphatics. For example, our laboratory has pioneered the use of intravital microscopy for investigating leukocyte migration and behaviour in afferent lymphatics with single-cell resolution. This work has revealed that lymphatic migration occurs in a step-wise manner: upon chemotactic guidance and entry into lymphatic capillaries, leukocytes initially actively crawl in lymphatic capillaries and frequently arrest to interact with LECs. Once the cells have arrived in contracting collecting vessel segments, the elevated lymph flow in this compartment allows for their detachment and passive, rapid transport to draining lymph nodes. Using time-lapse microscopy and other types of migration assays we have described several molecules involved in distinct steps of this migratory cascade. Performing bulk and single-cell RNA sequencing we are furthermore investigating subset-specific differences of LECs located in different segments of the lymphatic vessel tree, and how the LEC phenotype is impacted by lymph flow, inflammatory conditions or tumor growth.
By deciphering the mechanisms of lymphatic migration and the immunomodulatory properties of LECs, we aim to advance our basic understanding of these processes and to identify new therapeutic targets for the treatment of immune-mediated disorders. While we traditionally have mainly worked in vitro and in vivo in murine models, collaborations with clinical partners from the skintegrity.ch network have now allowed us to expand and complement our research by studies performed in human skin.
Selected publications
SKINTEGRITY.CH Principal Investigators are in bold:
- Arasa J, Collado-Diaz V, Kritikos I, Medina-Sanchez JD, Friess MC, Sigmund EC, Schineis P, Hunter MC, Tacconi C, Paterson N, Nagasawa T, Kiefer F, Makinen T, Detmar M, Moser M, Lämmermann T, Halin C (2021) Upregulation of VCAM-1 in lymphatic collectors supports dendritic cell entry and rapid migration to lymph nodes in inflammation. J Exp Med. 2021, 218(7):e20201413.
- Friess MC, Kritikos I, Schineis P, Medina-Sanchez JD, Gkountidi AO, Vallone A, Sigmund EC, Schwitter C, Vranova M, Matti C, Arasa J, Saygili Demir C, Bovay E, Proulx ST, Tomura M, Rot A, Legler DF, Petrova TV, Halin C. (2022). Mechanosensitive ACKR4 scavenges CCR7 chemokines to facilitate T cell de-adhesion and passive transport by flow in inflamed afferent lymphatics. Cell Rep. 38(5):110334.
- Montoya-Zegarra JA, Russo E, Runge P, Jadhav M, Willrodt AH, Stoma S, Nørrelykke SF, Detmar M, Halin C. (2019). AutoTube: a novel software for the automated morphometric analysis of vascular networks in tissues. Angiogenesis. 22:223-236.
- Vranova M, Friess MC, Haghayegh Jahromi N, Collado-Diaz V, Vallone A, Hagedorn O, Jadhav M, Willrodt AH, Polomska A, Leroux JC, Proulx ST, and Halin C (2019). Opposing roles of endothelial and leukocyte-expressed IL-7Rα in the regulation of psoriasis-like skin inflammation. Sci Rep., 9, 11714.
- Willrodt AH, Beffinger M, Vranova M, Protsyuk k D, Schuler K, Jadhav M, Heikenwalder M, van den Broek M, Borsig L, and Halin C (2017). Stromal expression of activated leukocyte cell adhesion molecule (ALCAM) promotes lung tumor growth and metastasis. Am J Pathol., 187, 2558.
